Below you’ll find answers to our frequently asked questions. If you have any other questions, please contact us at covid19@delft.care
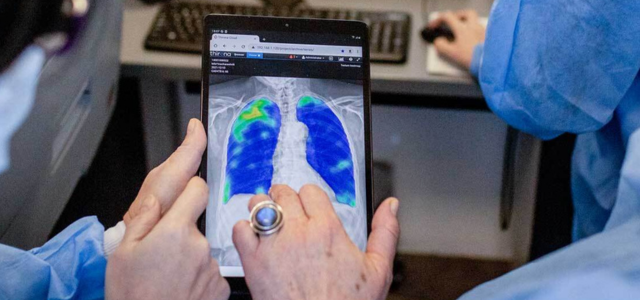
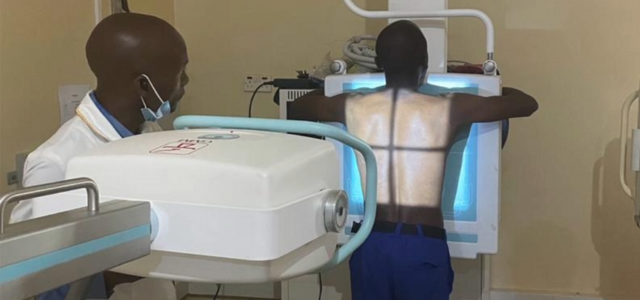
CAD4COVID processes CXR (chest X-ray images, both from CR and DR X-ray) in DICOM format of suspected COVID-19 cases.
Yes, CAD4COVID is a CE certified tool. Our Notified Body has granted CE certifications for both our CAD4COVID-CT and CAD4COVID-XRay solutions. Both have been granted Class II CE certifications, meaning they can officially be used as tools to aid in the interpretation of imaging COVID-19. The CE (0344) approval will help to further support the roll-out of both solutions, which will continue to be available free-of-charge to support healthcare facilities globally to face the pandemic.
To make CAD4COVID available globally to all institutions and healthcare facilities who want to access it, we have made CAD4COVID available as a Cloud-based solution (for the first phase of implementation). Users will receive a unique account for their facility or organization, which will provide access to the CAD4COVID viewer. Here, images can be uploaded and the user will, within the viewer, directly see the results on their images.
Yes, but, to allow as many users as possible to access CAD4COVID as quickly as possible, we will focus on a Cloud integration first (first phase of implementation). However, there is already a plan in place for PACS integration; more information on this will follow soon. If you fill in the form, you will also receive future development updates.
At the moment, CAD4COVID is offered as a standalone (free-of-charge) AI module and not integrated within the existing CAD4TB software. Please fill in the form on the CAD4COVID page to stay informed on development updates regarding CAD4COVID.
No, CAD4COVID is intended to inform to support the triage of COVID-19 and not optimized for TB screening.
CAD4COVID is a free-of-charge solution and will improve with additional COVID-19 CXR images. However, we will process your DICOM images via the Cloud for the purpose of providing you with the CAD4COVID results. We will not use your data for product development purposes (for CAD4COVID or any other solutions) for other purposes, unless you have given your explicit permission via a Data Usage Agreement. We do strongly welcome partners that want to share their COVID-19 access and they will be granted access to CAD4COVID at a prioritized basis as the data shared will allow for further optimization of CAD4COVID, offering an improved solution for all organisations accessing it.
At the moment (November 2020), CAD4COVID-Xray and CAD4COVID-CT are available in over 120 hospitals and clinics across 40+ countries.